 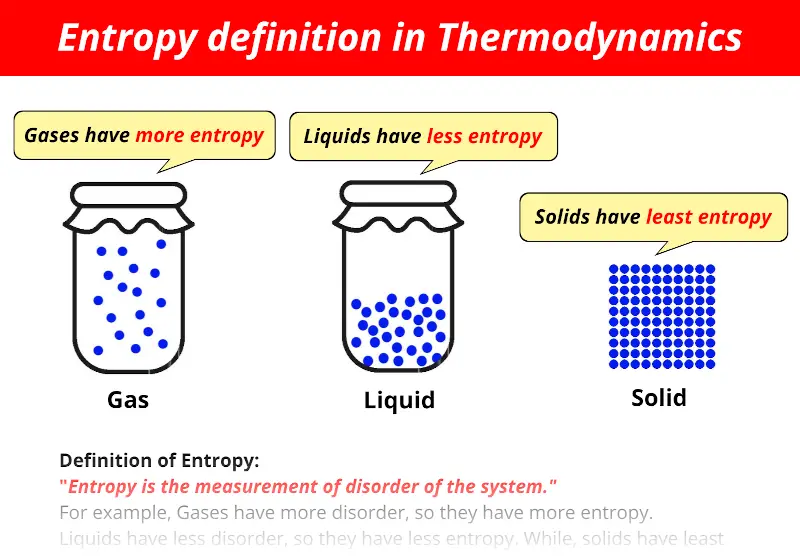
The process taken as a whole results in a net increase in disorder. Crystals are more orderly than salt molecules in solution however, vaporized water is much more disorderly than liquid water. In another example, crystals can form from a salt solution as the water is evaporated. Even when order is increased in a specific location, for example by the self-assembly of molecules to form a living organism, when you take the entire system including the environment into account, there is always a net increase in entropy. Mitra explained that all processes result in an increase in entropy. "At a very microscopic level, it simply says that if you have a system that is isolated, any natural process in that system progresses in the direction of increasing disorder, or entropy, of the system." "There are a number of ways to state the Second Law," Mitra told Live Science. This applies to literally every thing (every energy-involving activity or process) from biochemical reactions to electromagnetic radiation too (their particles are termed photons), and so it is a universal principle.Saibal Mitra, a professor of physics at Missouri State University, finds the Second Law to be the most interesting of the four laws of thermodynamics. You can only minimize this leakage by choosing a system that has the least possible degrees of freedom. He concluded that as long as the motor is made of material that consists of particles there will be leakage of some energy to the particles by means frictional dissipation and you cannot convince the particles not to do so. If your activity involves fancy properties (not so large number of particles but a highly flexible molecules in themselves, hence again higher degrees of freedom) again you have to pay a higher tax.Ĭarnot realized the concept of entropy when he was trying to design a perpetual motor. If your activity involves lots of properties (number of particles or large size of the system, therefore higher degrees of freedom) you have to pay a higher tax ( termed entropic penalty). You have to pay the tax (entropy) for every activity involving energy (money) other wise your activity will not be possible. If any system goes against this, it does so via an outside energy source which has increased the entropy of the universe somewhere else.Īs tax money is no longer available for you payments, entropy is the tax of nature in that it is the energy taken (leaked) from your system, be it supplied or produced, that is no longer available for work. In essence: things are always going towards a low energy state, i.e. So while the ATP synthase molecule is making higher energy molecules (decreasing entropy) it is doing so as H+ naturally flows towards equilibrium, which increases the overall entropy in the universe. It is, but it can only do so by the natural flow of H+ ions which have accumulated on one side of the cell membrane and naturally flow from their high concentration to their low concentration on the other side. This too seems like a system changing molecules from a low energy state to a high energy state. It is creating ATP (Adenine tri-phosphate) molecules from ADP (adenine di-phosphate) and a phosphate group. Think of the ATP Synthase on the cell membrane. Reactions which seem to increase the state of energy, or perhaps the 'order' of the universe, have done so at the expense of another part of the universe, usually through the conversion of energy from molecules such as ATP or NADPH. This becomes important in metabolic reactions. 
This can also be stated as the fact that the universe naturally falls towards the lowest energy state, or equilibrium. 2nd law of thermodynamics states that the overall entropy of the universe cannot decrease at any time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
